Spotlab
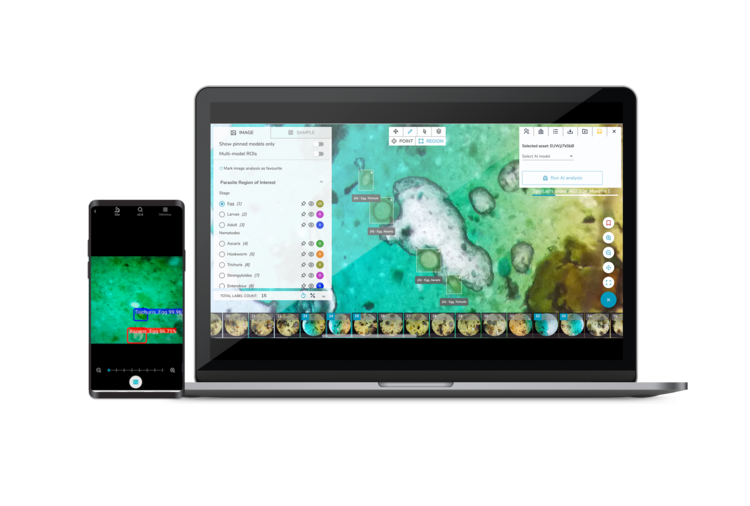
Spotlab is a social enterprise on a mission to use AI medical imaging diagnostics to democratise access to diagnosis, reducing the times, costs, distances and carbon footprint of existing imaging methods. Bringing software, an app-based interface and 3D printed hardware together, Spotlab has created an end-to-end diagnostic ecosystem, focused across a range of areas including infectious diseases, haematology and pathology.
We supported Spotlab in achieving CE marking for their combined IVD hardware and Software-as-Medical-Device offering.
XXX
What they needed
As a spin-out from the Polytechnic University of Madrid, Miguel and the Spotlab team initially began their work as a cutting-edge research project. As their research snowballed from a research project into a proof of concept, and by engaging with end-users, they realised the time had come to align with the appropriate quality and security standards. They quickly realised the risks and complexities of the regulatory frameworks and consequently approached Hardian Health for partnership and support.
Who are they?
Miguel Luengo-Oroz,
Co-founder, Spotlab:
“Regulation can be a huge barrier to entry for many startups in the medical device space due to the abundant complexities."


“Regulation can be a huge barrier to entry for many startups in the medical device space due to the abundant complexities.”
Miguel,
Co-founder, Spotlab
How we helped
With our more innovative clients who are breaking new ground, we often work together to fully understand the product whilst carving out a brand-new regulatory pathway that is fit for purpose and specific to the technology's use case and nuances. This was our approach with Spotlab. After much deliberation, we went back to basics and together took a stepwise approach, focusing first on achieving IVD regulation to lay the foundation for the submission and approval of the clinical algorithms. We worked with the Spotlab team to ensure that their processes are suitable for regulatory submission.
The result
Spotlab achieved CE marking for their software IVD. Building on those experiences, it has been quicker and easier for them to produce and receive approval on newer algorithms. Together, we have created a regulatory factory that complements and supports their AI factory, with structured and replicable parameters for how the data is created, how the algorithm is trained and how the bias of the algorithm is assessed, designed to carry all 105 of their identified disease algorithms through to approval.
