Envisonist Deep AI
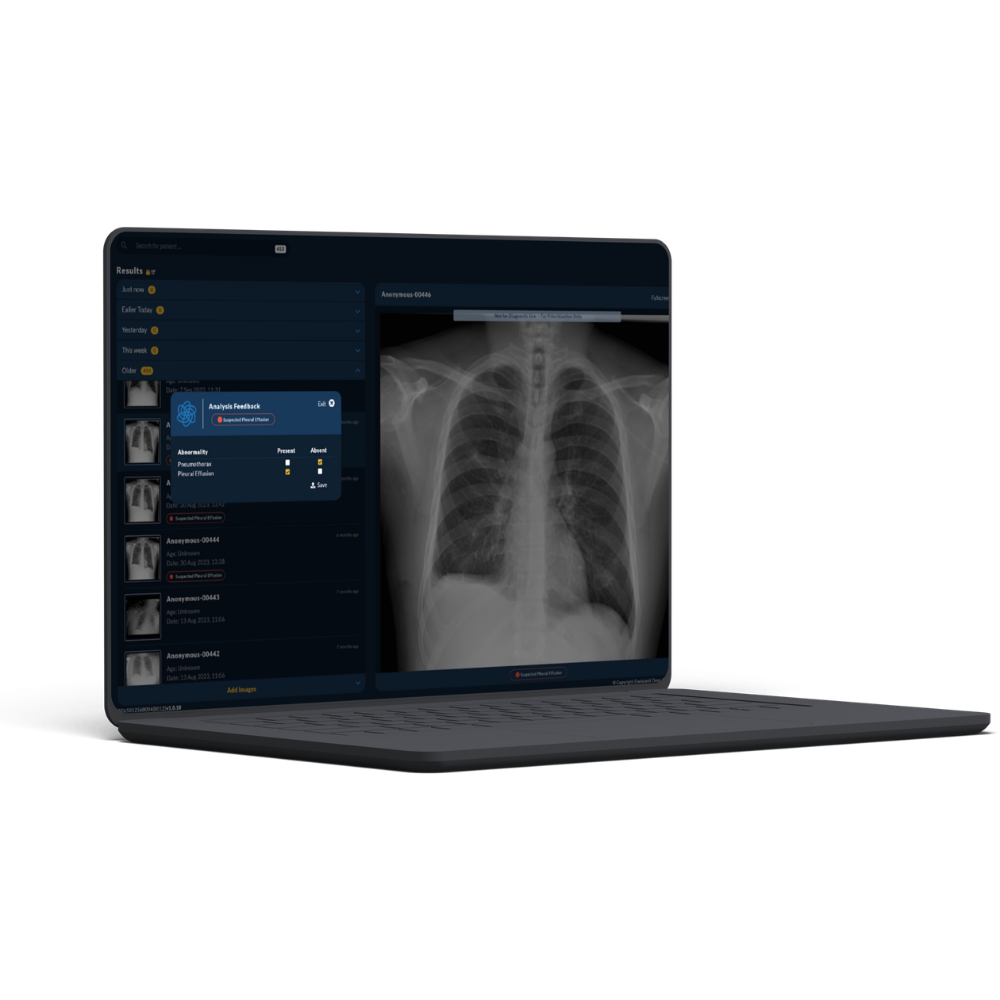
Envisionit Deep AI is an African-founded healthtech company developing AI-powered diagnostic tools to improve access to high-quality radiology services globally. The company’s mission is to democratise diagnostic healthcare, particularly in regions with limited access to specialist expertise. Envisionit’s assisted radiology solution, RADIFY® AII, supports emergency and intensive care teams by rapidly triaging radiology images in time-critical settings.
We supported Envisonist Deep AI in securing FDA 510(k) clearance for RADIFY® AI
FDA code: xxx
What they needed
Envisionit needed regulatory expertise to navigate the FDA 510(k) pathway for their AI-enabled medical device. This included defining a clear regulatory strategy, addressing evolving FDA cybersecurity requirements, and generating the robust clinical and technical evidence required to achieve clearance efficiently.
Who are they?
Dr. Jaishree Naidoo,
CEO, Envisionit Deep AI:
“The regulatory expertise of the Hardian Health team guided us in understanding the intricate details and requirements, as well as the compliance standards that were necessary for our product’s approval.”


“The regulatory expertise of the Hardian Health team guided us in understanding the intricate details and requirements, as well as the compliance standards that were necessary for our product’s approval.”
Dr. Jaishree Naidoo,
CEO, Envisionit Deep AI
How we helped
We worked closely with Envisionit to establish a clear Intended Use Statement and identify appropriate predicate devices. By building a fit-for-purpose Quality Management System we enabled early identification of operational gaps and strengthened long-term regulatory readiness. Our clinical and technical experts guided the validation strategy, supported the use of Envisionit’s proprietary RATify platform, and ensured clinical evidence met FDA expectations.
The result
RADIFY® AI achieved FDA 510(k) clearance, making Envisionit the first African-founded company to receive FDA clearance for an AI medical device. The FDA raised no findings on the cybersecurity approach taken, and Envisionit emerged with a strong regulatory and quality foundation to support future growth.
